8.3 Strategies and Mechanisms for Osmotic Stress Tolerance
KEY CONCEPTS
By the end of this section, you will be able to do the following:
- Explain how the main strategies cells can use to combat hypoosmotic and hyperosmotic stress are effective at restoring cell volume homeostasis.
- Give examples of two mechanisms that can be used to combat hyperosmotic stress, and organisms that use those mechanisms.
- Give examples to two mechanisms that can be used to combat hyperosmotic stress, and the organisms that use those mechanisms.
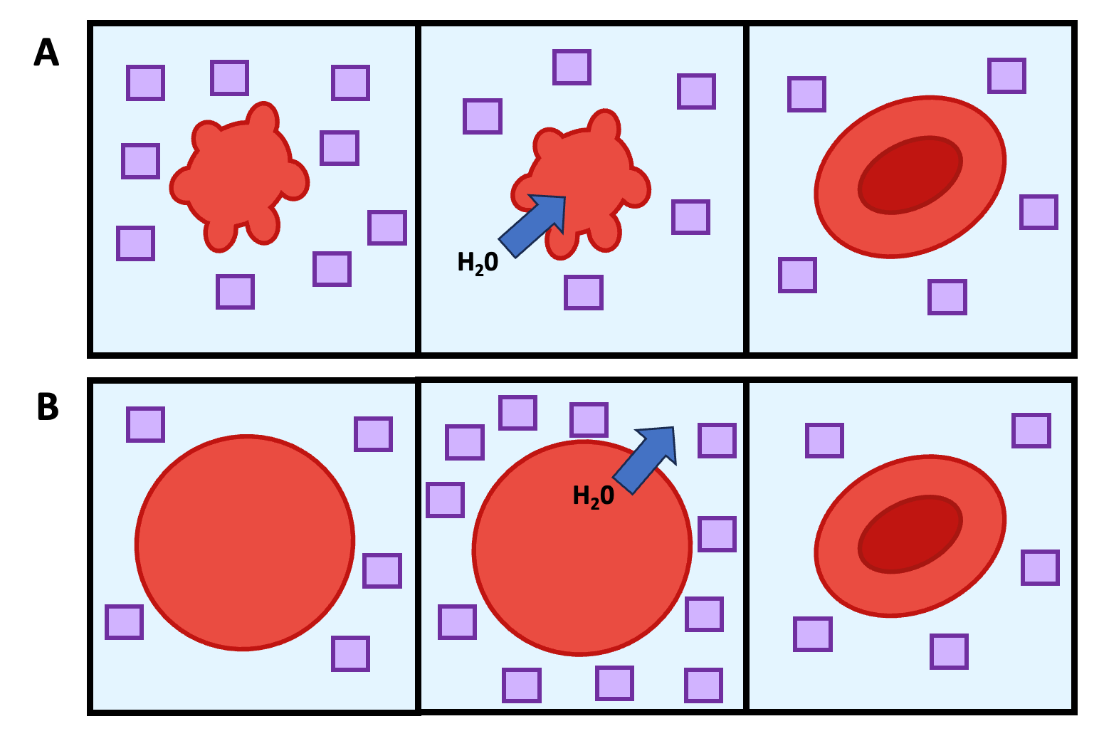
Once a cell has detected osmotic stress (Chapter 8.2), that cell must respond to restore normal function. The general strategies a cell uses to combat osmotic stress varies, depending on if the cell experiences a hyperosmotic or hypoosmotic environment. To counteract water loss during hyperosmotic stress, cells must use a strategy that causes water to re-enter the cell by osmosis (Figure 8.5). Meanwhile a hypoosmotic environment makes the cell swell as there are more solutes inside the cell. The cell responds by with strategies that cause water to leave the cell via osmosis (Figure 8.5). Cells can use a variety to mechanisms to enact these strategies; most of them involve modifying intracellular or extracellular solute concentrations to regulate water movement into or out of the cell via osmosis. Note that if you need a refresher on the difference between strategies and mechanisms, consult Chapter 7.
Mechanisms to Combat Hyperosmotic Stress
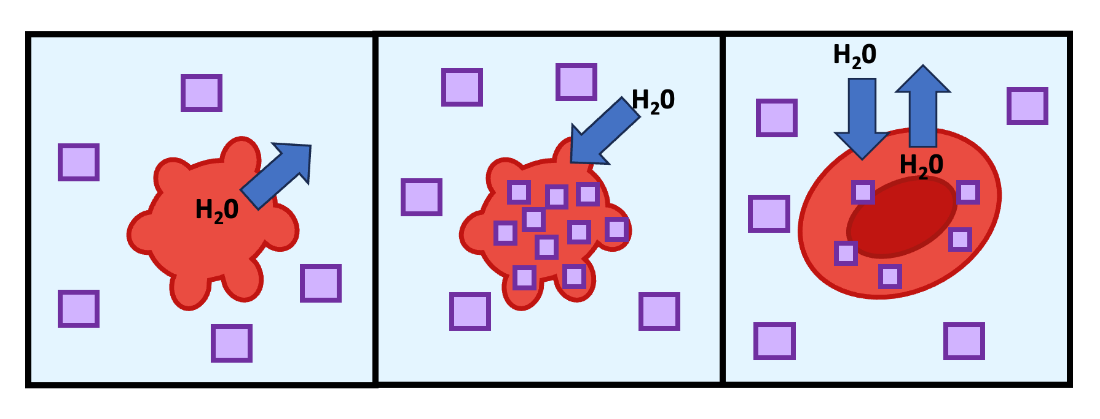
Cells under hyperosmotic stress can re-establish water balance using several mechanisms, usually involving an increase in intracellular solute concentration. Cells can either pump solutes into the cell from the extracellular environment or synthesize their own solutes internally (Figure 8.6). Compatible osmolytes are solutes that a cell can accumulate to high concentrations (to increase osmolarity) without harming the cell. Most solutes are not good compatible osmolytes; for example, it is dangerous for cells to accumulate high concentrations of ions. However, molecules like glycerol, some amino acids (e.g., proline) and some sugars (e.g., trehalose) can be accumulated to high levels without damaging the cells. For example, the unicellular algae Dunaliella salina live in salt water ponds that are hyperosmotic. These algae combat hyperosmotic stress by accumulating the compatible solute glycerol in their cytosol, which increases cytosolic osmolarity and helps prevent water loss. Similarly, the nematode, Caenorhabditis elegans, can also increase glycerol synthesis in order to increase intracellular osmolarity and prevent the sudden influx of water when the worm encounters a hyperosmotic environment.
Mechanisms to Combat Hypoosmotic Stress
As mentioned earlier, cells can mitigate potential damage when exposed to hypoosmotic environments by removing water from the cell. For example, freshwater protists such as Amoeba and Paramecium live in hypoosmotic environments. To prevent cellular lysing from an influx of water, these protists pump ions into their contractile vacuole – a structure indicated in both protists in Figure 8.7. Water follows into the hyperosmotic vacuole via osmosis. The contractile vacuole then expels the ions and water collected into the surrounding environment.
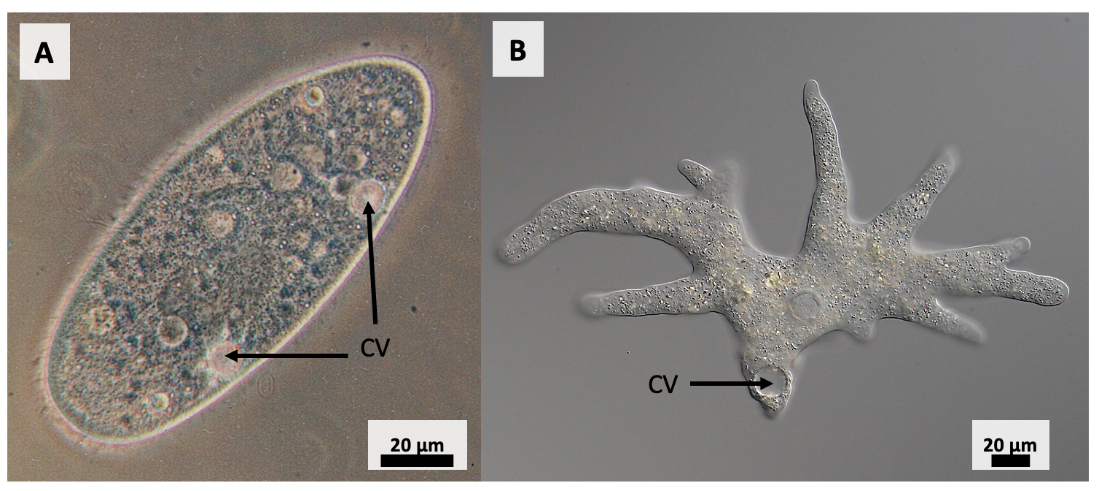
Another short-term response to hypoosmotic stress is to pump ions out of the cell, causing water to leave the cell via osmosis. This has been documented in hepatocytes (liver cells) of the turbot fish (Scophthalmus maximus) (Figure 8.8). A study by [1]Ollivier et al. revealed that the movement of K+ and Cl– out of the cell is directly involved in volume regulation during hypoosmotic stress. A combination of ion channels, including uniport K+, uniport Cl–, Cl–/HCO−3 exchangers, and K+/Cl– cotransporter channels are thought to be responsible for the movement of these ions during the response to osmotic shock. As K+ and Cl– leave the hepatocyte, water soon exits, decreasing cellular volume back to normal.

- Ollivier, H., Pichavant, K., Puill-Stephan, E., Roy, S., Calvès, P., Nonnotte, L., and Nonnotte, G. 2006. Volume regulation following hyposmotic shock in isolated turbot (Scophthalmus maximus) hepatocytes. J Comp Physiol B 176(5): 393–403. doi:10.1007/s00360-005-0061-8. ↵

